Phase 3 trial
DUVYZAT was studied in EPIDYS, one of the most inclusive phase 3 DMD studies to date1,*
EPIDYS was a global, 18-month, double-blind, randomized, placebo-controlled trial (N=179).1,2

Ethan, actual DUVYZAT patient for 5+ years.
- Ambulant boys aged ≥6 years with genetically confirmed DMD who were receiving a stable corticosteroid regimen (median age of 9.8 years)1,2
- All patients completed two 4SC assessments (mean of ≤8 seconds) and a time-to-rise assessment (≥3 but <10 seconds)1
- No restrictions on genetic mutations, but these were generally balanced across both groups1
Efficacy was assessed in 120 patients using multiple measures, including 4SC,† NSAA (including time-to-rise from the floor), 6-minute walk test, change in VLFF, and a muscle strength assessment (knee extension and elbow flexion).1
Safety was assessed in 179 patients (Group A + Group B).1
Efficacy and safety analysis, n=120
- Baseline VLFF >5% but ≤30%
- Unlikely to lose mobility abruptly but expected to show measurable decline in function, strength, and fat fraction on placebo + corticosteroids
Safety analysis only, n=59
- Baseline VLFF ≤5% or >30%
- Recruited to assess the safety of DUVYZAT + corticosteroids in a broader population of patients with DMD

Ethan, actual DUVYZAT patient for 5+ years.
PRIMARY ENDPOINT: 4SC
PROVEN PROTECTION against DMD progression with DUVYZAT2
Patients treated with DUVYZAT were able to complete 4SC faster than patients taking placebo2
Change in time to perform 4SC vs placebo2
KEY SECONDARY ENDPOINT: NSAA
DUVYZAT demonstrated greater preservation of motor function vs placebo as shown by NSAA1,§
Change from baseline in total NSAA item score over 18 months1,||
§ Nominally significant but not statistically significant based on prespecified multiplicity adjustment.1
II NSAA is a DMD-specific assessment scale measuring lower limb function in ambulant children with DMD comprising 17 items scored on a scale of 0 to 2. A score of 2 indicates the activity is performed without difficulty, 1 indicates the activity is performed with some compensation, and 0 indicates the activity cannot be performed independently.1,2,5
KEY SECONDARY ENDPOINT: VLFF
Compared with placebo at 18 months,
DUVYZAT reduced new fat infiltration in key muscle groups required for ambulation1,2,6,7,§
Change in mean VLFF over 18 months from baseline1,6
At 18 months, for patients with baseline VLFF in the range of >5% to ≤30%, mean increase (absolute difference from baseline) in VLFF was 7.48% in DUVYZAT-treated patients compared to 10.89% in patients who received placebo.2
§ Nominally significant but not statistically significant based on prespecified multiplicity adjustment.1
EXPLORATORY ANALYSIS: VLFF
DUVYZAT reduced new fat infiltration in the thigh muscle1,2,6
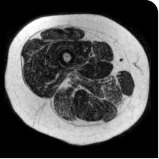
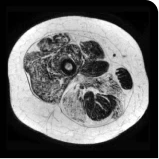
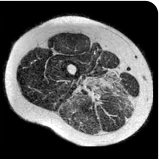
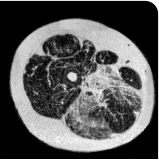
MRS images showing VLFF increase from baseline after 18 months.1
Images are from single individuals and may not be representative of all patients' results.
MRS, magnetic resonance spectroscopy.
DUVYZAT safety
DUVYZAT safety is based on the pivotal EPIDYS 18-month study in a total of 179 ambulant patients with DMD aged 6 years and older on concomitant corticosteroid treatment.2
Adverse reactions reported in >5% of DUVYZAT-treated patients2
| Adverse reaction, % | DUVYZAT + corticosteroids (n=118) | Placebo + corticosteroids (n=61) |
|---|---|---|
| Diarrhea | 37 | 20 |
| Abdominal pain | 34 | 25 |
| Thrombocytopenia‡ | 33 | 0 |
| Nausea/vomiting | 32 | 18 |
| Hypertriglyceridemia | 23 | 7 |
| Pyrexia | 13 | 8 |
| Myalgia | 9 | 3 |
| Rash | 9 | 2 |
| Arthralgia | 8 | 2 |
| Fatigue | 8 | 0 |
| Constipation | 7 | 2 |
| Decreased appetite | 7 | 0 |
-
Diarrhea, the most common adverse reaction, usually occurred within the first few weeks of treatment and resolved with continued dosing2,4
- Antiemetics or antidiarrheal medications may be considered during treatment with DUVYZAT. Fluid and electrolytes should be replaced as needed to prevent dehydration2
‡ Thrombocytopenia includes platelet count decreased and thrombocytopenia.
References: 1. Mercuri E, Vilchez JJ, Boespflug-Tanguy O, et al; EPIDYS Study Group. Safety and efficacy of givinostat in boys with Duchenne muscular dystrophy (EPIDYS): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2024;23(4):393-403. 2. DUVYZAT. Prescribing information. ITF Therapeutics; 2024. 3. Muntoni F, Signorovitch J, Sajeev G, et al. Meaningful changes in motor function in Duchenne muscular dystrophy (DMD): a multi-center study. PLoS One. 2024;19(7):e0304984. 4. Data on file. ITF Therapeutics; 2025. 5. Ayyar Gupta V, Pitchforth JM, Domingos J, et al. Determining minimal clinically important differences in the North Star Ambulatory Assessment (NSAA) for patients with Duchenne muscular dystrophy. PLoS One. 2023;18(4):e0283669. 6. Vandenborne K. Givinostat in DMD: results of the Epidys study with particular attention to MR measures of muscle fat fraction. Oral presentation at: Muscular Dystrophy Association Clinical & Scientific Conference; March 19-22, 2023; Dallas, TX. 7. Liu MQ, Anderson FC, Schwartz MH, Delp SL. Muscle contributions to support and progression over a range of walking speeds. J Biomech. 2008;41(15):3243-3252.
